 You will also need the card in the upper right corner. For this lesson you will need the 5 cards for each element from the left side of each sheet. Laminate each page and cut out the cards. About this Lessonīoth this lesson and Lesson 4.3-The Periodic Table and Energy Level Models both use the atom description cards. The activity sheet will serve as the “Evaluate” component of each 5-E lesson plan. 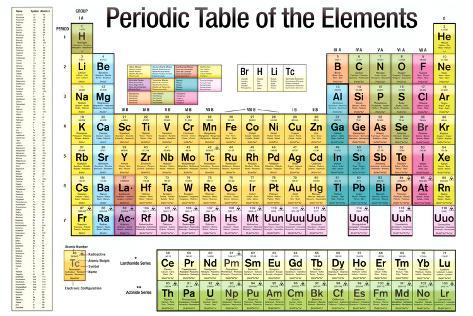
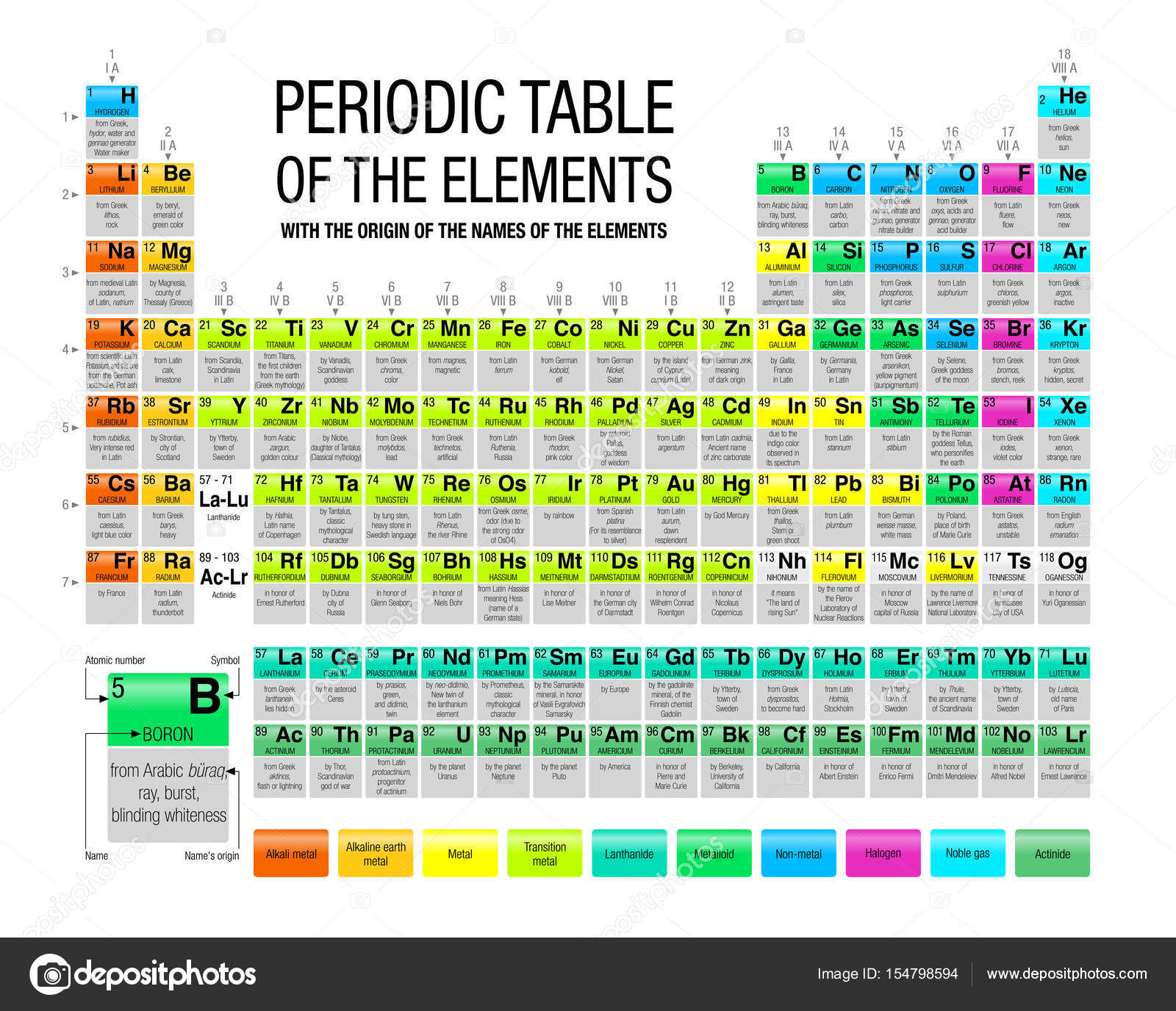
Evaluationĭownload the student activity sheet, and distribute one per student when specified in the activity. They will also be able to explain the meaning of atomic number and atomic mass. Students will identify different atoms by the number of protons in the nucleus and realize that the number of electrons equals the number of protons in a neutral atom. Students will then watch several videos of some interesting chemical reactions involving some of these elements. They will try to correctly match cards with information about an element to each of the first 20 elements. Students will focus on the first 20 elements. They will be introduced to the basic information given for the elements in most periodic tables: the name, symbol, atomic number, and atomic mass for each element. Students will begin to look closely at the periodic table. The atoms in the periodic table are arranged to show characteristics and relationships between atoms and groups of atoms.The atomic mass of an element is the average mass of the different isotopes of the element.Atoms of the same element with different numbers of neutrons are called “isotopes” of that element.Different atoms of the same element can have a different number of neutrons.The number of electrons surrounding the nucleus of an atom is equal to the number of protons in its nucleus.The atomic number of an atom is equal to the number of protons in its nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
